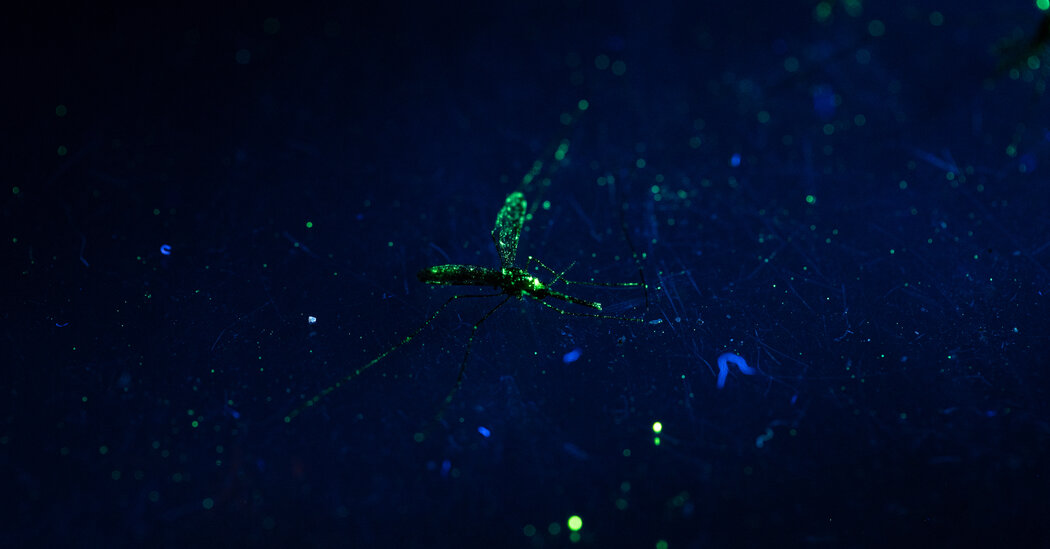
On a muggy evening in July on the island of Príncipe, part of a volcanic archipelago 200 miles off the West African mainland, 11,000 mosquitoes dusted in fluorescent green powder flew together into the heavy equatorial air, tiny volunteers in the service of science.
Over the next 10 nights, another group of volunteers, human ones, sat outside their houses in villages nestled in the rainforest, keeping their arms and legs exposed in the damp dark, waiting for the faint tickle of a mosquito in search of blood. Once one alighted, they switched on a headlamp and used a rubber tube attached to a glass vial to suck the insect up and seal it in a cup.
The mosquitoes were raised from larvae, dusted green, then set free, by an international team of scientists who are trying to bring cutting-edge genetic science to an ancient fight — that against malaria, the most deadly mosquito-borne disease.
For each of the 10 mornings after the mosquito release, the scientists fanned out along the northeastern coast of this remote island, collecting cups humming with mosquitoes. They then took the insects to a makeshift lab in their hotel suite in the island’s one town, Santo Antonio, where they slid them under the light of a fluorescent microscope. Twelve of the 253 mosquitoes that had been caught glimmered with tiny particles of the green powder that clung to their scaly bodies.
The recaptured green mosquitoes offered insight into how far they flew and the size of the mosquito population, clues to the dynamics of malaria in this country. And they moved the scientists one step closer to their goal: replacing the mosquitoes that live here now with ones they have genetically modified so that they can no longer transmit the malaria parasite.
Their idea is to release a small colony of genetically modified mosquitoes, just the way they did with the green-dusted ones, to mate with wild ones. The gene engineering technology they are using could, in just a few generations — a matter of months when it comes to mosquitoes — make every member of the species that transmits malaria here, the Anopheles coluzzii, effectively immune to the parasite.
This team, working with a project called the University of California Malaria Initiative, has already successfully engineered the Anopheles coluzzii to block the parasite in a lab. And the scientists believe they can harness gene drive, a process in which an inherited trait spreads swiftly throughout a population, so that all the species’s offspring will carry it, not just half, which is the way inheritance normally works.
The malaria situation in São Tomé and Príncipe, an African island nation with a population of 200,000, epitomizes the current challenge in the global struggle against the disease. The country is among the world’s least developed, and it has depended on foreign aid to fight malaria. Various campaigns over the past 50 years drove cases down, only to have them resurge worse than ever when the benefactor moved on.
Over the past 18 years, with nearly $21 million from the Global Fund to Fight AIDS, Tuberculosis and Malaria, São Tomé has used a package of tools — including insecticide-treated bed nets; new and better drugs; killing larvae in bodies of water; and indoor spraying of homes — to stunning effect. No one has died of malaria here in the past five years.
Still, there were 2,000 cases of malaria here last year, and the disease can be fatal and create serious economic burdens. The country, much of which is a pristine protected biosphere rich in birds and orchids, depends heavily on tourism from Europe. Being certified as malaria-free would be a huge benefit.
The journey from a few thousand cases to elimination is complicated and costly; some experts say it’s as hard to close that last gap as it is to eliminate the first 90 percent of cases. Here, as in other countries that have dramatically suppressed malaria, mosquitoes have evolved to resist all of the insecticides now in use. They have started to bite outdoors and in the daytime, when people are not under bed nets, instead of indoors and at night, when most malaria transmission used to occur. The parasite itself is evolving to resist the main treatments. And malaria funding has plateaued even as the necessary interventions have grown more costly.
These countries need a way to fight the disease that is permanent and does not require continuous investment.
Greg Lanzaro, a molecular geneticist at the University of California, Davis, who leads the malaria team, believes his group has that solution.
“We’ve been working on this for 30 years, and from the beginning we said, ‘It has to work, but it also has to be inexpensive, and it has to be sustainable,’” he said as he watched the mosquitoes being released in a Santo Antonio park. “And we believe we have it.”
But genetic modification is a controversial endeavor. Governments are hesitant, and few in Africa have laws to regulate the use of the technology. Its risks lie in the unknowns: Could the modified mosquito evolve in some way that has harmful effects on the rest of the ecosystem? Could it prompt a dangerous mutation in the malaria parasite, which will find a new way to spread to survive?
It is, in essence, the Jurassic Park question: Could meddling in genetic code have catastrophic consequences that no one anticipates? (The original Jurassic Park experiments were carried out on a remote tropical island to minimize risk. We know how that turned out.)
Yata Mota, who works as a guide at a tourist center on a coffee plantation on São Tomé, said she was initially hesitant when she heard about the genetic modification proposal. “We would be the first place in the world with these mosquitoes, and that scares me: When it’s the first time they’re doing it, you don’t know what could happen,” she said.
These fears are why the University of California team chose São Tomé and Príncipe for its experiment: The island nation is isolated and has limited international traffic. The team has also built in a plan to wipe out the population of its modified mosquitoes if there is a need to end the experiment for any reason.
There are a handful of other projects working on ways to use genetic modification against malaria and other mosquito-borne diseases in Africa and beyond. Some have tried spreading a genetic trait by releasing millions of mosquitoes into a wild population — the “inundation” technique. The logistics of that approach have always seemed improbable to the California team, because they require continuous releases of insects and huge investments in infrastructure.
A gene drive, however, is an efficient way to spread a protective modification through a wild mosquito population, fast. “You’re letting the mosquito do the work for you,” Dr. Lanzaro explained.
African opponents of genetic modification say it is neither well-enough understood to be safe, nor necessary. “The provision of basic sanitation and better and safer housing would not only eradicate the disease, it would boost the local economy,” said Nnimmo Bassey, a prominent Nigerian environmentalist.
“The problem we have seen here is that the agency and the scientists cannot explain the nature of the genetically modified organisms or the implications of releasing them into the population in ways that people understand,” said Mr. Bassey, who heads the Health of Mother Earth Foundation. “People cannot consent to what they do not understand. They’re just being used as guinea pigs.”
Abdoulaye Diabaté, who runs the most advanced mosquito genetics program in Africa, said he understood these concerns but argued that anxiety was an insufficient reason not to try genetic modification.
“We may not know what may happen but we know what is happening today: 600,000 people dying of malaria, and we need to fix it,” said Dr. Diabaté, the principal investigator in Burkina Faso for Target Malaria, a project backed by the Bill & Melinda Gates Foundation. “We can’t say we are afraid of the future so we will accept 600,000 people dying. We make good progress as a society when we invest in our dreams, rather than our fear.”
Unlike other mosquito-control efforts, he said, genetic modification promises to benefit everyone in a community equally, regardless of income level, which is not the case with products like bed nets, insecticides and vaccines. And while people from high-income nations express fears about genetic modification, Dr. Diabaté hears far fewer of these concerns from people in villages whose children get malaria six or eight times a year, he said.
The University of California project has poured resources and energy into public education to advertise the benefits of genetic modification and try to assuage fears; it has teams of community agents educated on the gene drive process who can field questions, and it presents to school groups across São Tomé and Príncipe. In an effort to avoid the perception that foreign scientists are experimenting on the local population, the program has set up a laboratory at the University of São Tomé and Príncipe, where the genetic modification work would take place, and is funding graduate studies for São Tomense students.
But the program needs government approval to move forward with the genetic portion of the intervention and São Tomé and Príncipe, like many other African countries, does not yet have a legal framework for the use of genetically modified organisms. Legislation to establish one has stalled in the National Assembly. Without a body assessing the risks and safety of using a tool like these mosquitoes, the California team has no one to submit its project proposal to and is effectively stalled.
The country’s health minister, Celsio Junqueira, said in an interview that his government was focused on basic services such as getting water and electricity into primary care facilities, and that genetically modified mosquitoes were a luxury his government couldn’t spare time or energy on now. He did not have a timeline for when the University of California project might be able to proceed with actual, island-bred mosquitoes.
In 2021, the World Health Organization issued new guidelines to help countries think through how they can test mosquito-control interventions based on genetic modification, which the organization said could have potential as self-sustaining solutions, particularly when low case numbers create pressure to allocate resources away from malaria.
Ricarda Steinbrecher, a molecular geneticist who sits on the United Nations advisory council on synthetic biology, said that genetic modification projects required international regulation. “Mosquitoes cross boundaries, you cannot make them stop at the national borders,” she said.
This is one of the challenges for Target Malaria’s project: How can it keep its modified mosquitoes inside the borders of Burkina Faso? Target Malaria is modifying insects not to block the parasite, but essentially to wipe themselves out. The project has a genetically modified line of mosquitoes that produces sterile females, and another in which males are modified to produce predominantly male offspring, gradually distorting the population (only females bite and spread disease). This approach works to fight dengue fever and other kinds of mosquito-borne disease, not just malaria, since it eliminates the vector.
In 2019, Dr. Diabaté oversaw a small release of sterile males, the first field experiment in Africa with genetically modified mosquitoes, which was intended as an initial demonstration of safety. A British biotechnology company called Oxitec has a modified Aedes aegypti mosquito that it has been selling directly to consumers in Brazil since 2018, to fight diseases such as dengue and yellow fever. For the last two years, Oxitec has been releasing its modified mosquitoes in the Florida Keys and in California, after winning approval from the U.S. Environmental Protection Agency. That is a test to fight dengue; cases of the debilitating and sometimes fatal disease are climbing quickly as the warming climate expands the range of the mosquito that carries the illness.
Oxitec recently set up shop in Djibouti, on the Horn of Africa, where it is modifying Anopheles stephensi, a newly invasive species of mosquito that has caused a resurgence of malaria in a country that had almost eliminated it.
The Oxitec and Target Malaria approaches both take the mosquito out of the ecosystem. Proponents say this isn’t a problem — there are 3,500 species of mosquitoes in the world, and no animal that depends solely on one mosquito species for food. But the premise often makes people nervous.
The University of California approach carries less risk, and less of the appearance of meddling with nature, said Arlindo Carvalho, a former health minister of São Tomé and Príncipe who now advises various malaria-control projects including this one.
“Not eradicating, but modifying — this is the most secure and sustainable path.” The modification approach can also work on multiple diseases and species. And it doesn’t require the repeated release of massive numbers of mosquitoes, or the infrastructure to breed and rear them.
The genetic modification that the California team is proposing will be susceptible to the same evolutionary pressures as every other mosquito intervention: That is, nature will find a way around the modification the same way mosquitoes develop resistance to insecticides. The malaria parasite will eventually develop resistance to get around the modification making the mosquito immune.
“That’s the price of doing business,” said Dr. Lanzaro.
But he said the project is prepared for it. First, its modification attacks plasmodium falciparum, the most common and most lethal species of the malaria parasite, in two different ways — which makes it more difficult for it to develop resistance. The project has also developed modifications that focus on mosquito genes that trigger different immune responses to the parasite. “We have those on the shelves, ready to plug in,” he said. If the parasite started to show resistance — if malaria came back — the scientists in São Tomé could raise a colony of mosquitoes with the new modification swapped in, and release those, he said.
But until São Tomé has a governing body to whom the team can submit the reams of data it has amassed while studying the country’s ecology and mosquitoes — like the green ones the project tracked across Príncipe — it has no way of knowing if its method will work in the wild. A lab-bred, lab-tested mosquito is not a wild one. Will the team’s modified ones be appealing to potential wild mates? Will they be as successful in finding food and shelter? The team has no way of knowing.
“We have got to get going,” Dr. Lanzaro said. “We can’t just keep saying 10 more years, 10 more years. Six million people have died while we’ve been fiddling around.”